Renal cell carcinoma
In 1 minute
Renal cell carcinoma (RCC) is not just a generic name for any "regular" renal tumor. It is the main malignant epithelial tumor family radiologists worry about when an adult has a suspicious enhancing solid renal mass.2 Radiologists often start from the phrase enhancing renal mass because true enhancement means the lesion is behaving like solid tissue rather than a simple cyst, even though some small solid masses still turn out to be benign.13
The main imaging question is not just "is this a renal mass?" but "what kind of solid renal mass behavior is this?" Clear cell RCC usually enhances avidly in the corticomedullary phase, papillary RCC is more often low-enhancing and can look deceptively dense or pseudocystic, and chromophobe RCC often lands in between.24 Benign mimics matter because angiomyolipoma (AML) may show visible fat or, if fat-poor, overlap with RCC, and oncocytoma can also be hypervascular and sharply marginated.53
Fast engineer rule: enhancement + fat + T2 pattern + phase behavior matter more than the words "renal tumor" alone. Think stronger RCC concern when the lesion is a true enhancing solid mass without benign fat, especially if it looks heterogeneous, invasive, or subtype-typical. Slow down when the mass has macroscopic fat, a strong fat-poor AML pattern, or oncocytoma-like enhancement, because those are exactly where pathology labels and imaging impressions can diverge.159
Quick diagnostic logic
| Situation | What radiologists look for | Why it matters |
|---|---|---|
| Incidental solid renal mass on CT1 | True enhancement, whether the lesion is genuinely solid rather than cystic, visible macroscopic fat, and whether MRI is needed for a small or hypovascular mass | Enhancement starts the malignant workup, but small solid masses still have a meaningful benign fraction |
| Clear cell-leaning pattern24 | Avid corticomedullary hyperenhancement, heterogeneity, necrosis, washout, pseudocapsule, or venous invasion | This is the classic aggressive-looking RCC pattern and the subtype most people picture first |
| Papillary-leaning pattern25 | Homogeneous low-level enhancement, T2 hypointensity, precontrast hyperattenuation, or a pseudocystic look | Papillary RCC can look less dramatic than clear cell RCC and is easier to confuse with hemorrhagic cyst or fat-poor AML |
| Chromophobe or indeterminate solid RCC pattern24 | Intermediate enhancement between clear cell and papillary, relatively homogeneous appearance, and sometimes a central scar | Chromophobe RCC often overlaps with oncocytoma, so imaging may stay suggestive rather than definitive |
| AML or fat-containing mimic5 | Macroscopic fat below -10 HU, India-ink artifact, T2-dark fat-poor AML pattern, angular interface, and enhancement behavior | A benign AML can look like RCC if visible fat is absent, so the fat question is not all-or-none |
| Oncocytoma or benign-solid mimic problem39 | Homogeneous nephrographic enhancement, central scar, segmental enhancement inversion, and lack of malignant pseudocapsule clues | Oncocytoma is a major reason not every enhancing solid renal mass can be called RCC from imaging alone |
Example figures
These embedded examples are lesion-specific RCC subtype figures from an open access review distributed under the Creative Commons Attribution License.2 They are here to show the two ends of a common subtype contrast: classic hypervascular clear cell RCC versus lower-enhancement papillary RCC.
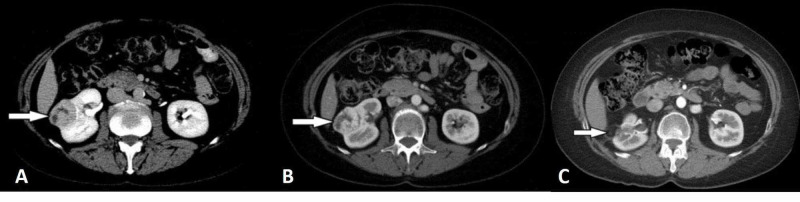
Figure 1. Clear cell RCC example from the source review. The corticomedullary and excretory phase CT images show the classic hypervascular pattern with later washout that makes radiologists lean toward clear cell RCC rather than papillary RCC. Source:
Morshid et al., Figure 1
, Creative Commons Attribution License.2
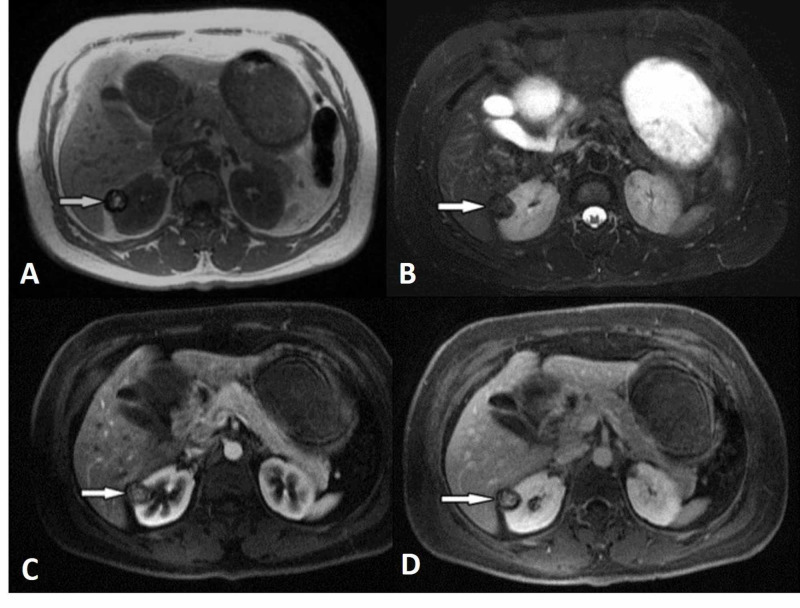
Figure 2. Papillary RCC example from the same review. The lesion is well-demarcated, hypovascular, and T2-dark, which is why papillary RCC can feel much less dramatic than clear cell RCC and sometimes drift toward cyst or fat-poor AML confusion. Source:
Morshid et al., Figure 4
, Creative Commons Attribution License.2
Additional link-only mimic examples:
- Benign oncocytoma example with a small hypervascular lesion and central scar: See original Figure 2.3
- Fat-poor AML example showing T2-dark signal and early enhancement with washout: See original Figure 5.5
Common confusions
- Angiomyolipoma (AML): classic AML becomes easier when gross fat is visible, but fat-poor AML is a real mimic problem. It may be hyperattenuating on unenhanced CT, T2-dark on MRI, and show early enhancement with washout, which means it can overlap with both papillary RCC and clear-cell RCC enhancement logic when the fat clue disappears.5
- Oncocytoma: this benign epithelial tumor can be sharply marginated, hypervascular, and sometimes show a central scar or segmental enhancement inversion. Those clues are helpful, but not reliable enough to exclude RCC, which is why oncocytoma remains one of the most important reasons an enhancing solid renal mass is not automatically malignant on imaging alone.39
- Hemorrhagic or proteinaceous cyst: this confusion matters most when papillary RCC is dense on precontrast CT or looks partly pseudocystic. A dense or T1-bright lesion can still be benign cystic content, but once the lesion behaves like a solid enhancing mass, the cyst explanation weakens fast.2 See also hemorrhagic renal cyst.
- Complex cystic RCC versus solid RCC: cystic RCC is driven by enhancing wall, septa, or nodules, whereas solid RCC is driven by enhancing soft tissue. Both may still be reported broadly as a "renal mass," so the engineer should preserve whether the lesion was fundamentally solid, cystic, or mixed.1
- Metastasis or lymphoma: these are secondary mimics rather than the first default explanation for an incidental cortical enhancing renal mass. They matter more when the pattern is multifocal, infiltrative, bilateral, or the patient history already points strongly away from a primary RCC story.3
Engineer-first takeaway
If you only remember one workflow rule, remember this: keep the renal mass phases together. Noncontrast, corticomedullary, nephrographic, excretory, and MRI problem-solving sequences are not redundant. They are the reason radiologists can say "clear cell-leaning," "papillary-leaning," "possible AML," or "still could be oncocytoma" instead of collapsing everything into a single vague tumor label.24
For informatics and AI work, it helps to treat enhancing renal mass as a stronger signal than the phrase renal tumor alone. The first phrase usually means the radiologist has already committed to a solid enhancing lesion, while the second may still hide whether the mass was cystic, fat-containing, subtype-leaning, or indeterminate. Good labels and NLP pipelines should keep nearby distinctions such as:
- enhancing solid renal mass
- cystic renal mass
- clear cell RCC
- papillary RCC
- chromophobe RCC
- angiomyolipoma
- oncocytoma
Public data can help, but the scope matters. KiTS23 is excellent for kidney, tumor, and cyst segmentation, yet it does not solve pathology subtype labeling. UCSF RMaC is closer to the subtype and enhancement problem because it includes multiphase CT and pathology, but it is still a broader renal-mass resource rather than a pure RCC-only benchmark.78 That gap between segmentation labels and subtype-aware diagnosis is an EnginRad inference from the dataset scope rather than a direct claim from either source.
Related research map
Start here
Clinical decision frame
Use this path if you want the shortest route from incidental enhancing renal mass to the main RCC versus mimic decision logic.
ACR incidental renal mass white paper -> Multimodality RCC review -> Small renal mass CT review
Subtype problem
Which subtype is this leaning toward?
Use this path when the real question is clear cell versus papillary versus chromophobe, and where oncocytoma still gets in the way.
Multimodality RCC review -> Multiphasic CT subtype paper -> RCC versus oncocytoma model validation
Engineer view
Data and workflow path
Use this path when you care about kidney lesion AI, segmentation resources, and the gap between pathology nuance and public labels.
Kidney lesion AI review -> KiTS23 -> UCSF RMaC
This is a curated research map, not an automatic similarity graph. It starts from the adult enhancing renal mass problem and then branches into subtype imaging papers, benign-mimic differentiation work, and engineering datasets.
This is a curated research map, not an automatic similarity graph. It starts from the adult enhancing renal mass problem and then branches into subtype imaging papers, benign-mimic differentiation work, and engineering datasets.
Guideline - 2017
ACR incidental renal mass white paper
This is the practical management anchor for incidental renal masses on CT.
Best for
Enhancement thresholds, why MRI is preferred for suspected hypovascular small masses, and why small solid masses are not automatically malignant.
Watch out
It is an incidental-mass management paper, not a subtype-imaging paper, so it frames the problem better than it solves clear cell versus papillary nuances.
Review - 2021
Multimodality RCC review
A concise, visual review of how RCC subtypes behave across the main imaging modalities.
Best for
Fast orientation to clear cell hypervascularity, papillary hypovascularity, chromophobe intermediate behavior, and reusable teaching figures.
Watch out
It is broad and concise, so it is stronger for pattern recognition than for the fine print of mimic management.
Review - 2023
Small renal mass CT review
Places RCC, AML, and oncocytoma in the same practical small-mass differential.
Best for
The real mimic problem: why benign solid renal masses still get resected, which CT clues help, and where the overlap remains stubborn.
Watch out
It is a review that summarizes mixed literature, so it should not be treated like a single definitive rule set.
Differentiation - 2013
Multiphasic CT subtype discrimination
A focused subtype paper on how clear cell RCC separates from other RCC subtypes and oncocytoma at multiphasic CT.
Best for
Learning that clear cell and oncocytoma often peak in the corticomedullary phase, while papillary and chromophobe more often peak later.
Watch out
It is a subtype-discrimination study in selected lesions, not a universal bedside rule for every renal mass workup.
Differentiation - 2022
RCC versus oncocytoma model validation
Directly tackles one of the hardest benign solid mimics: oncocytoma versus RCC.
Best for
Seeing which CT features stayed useful in validation, including homogeneous nephrographic enhancement and excretory-phase behavior.
Watch out
It validates a predictive model rather than creating a simple always-works radiology sign, so overlap still remains.
AI / Radiomics - 2023
Kidney lesion AI and radiomics review
Shows where renal lesion AI is trying to separate benign from malignant masses and subtype RCC before surgery.
Best for
A landscape view of AML-versus-RCC, oncocytoma-versus-RCC, subtype prediction, and where radiomics claims are strongest or still immature.
Watch out
It is a broad review, so it gives context well but does not solve a single lesion question by itself.
Dataset - 2023
KiTS23
The main public kidney CT segmentation benchmark that includes kidney, tumor, and cyst labels.
Best for
Segmentation pipelines, lesion localization, and engineering work where anatomy plus mass masks matter more than pathology subtype labels.
Watch out
It is a segmentation benchmark, not an RCC subtype dataset, so it does not tell you clear cell versus papillary versus benign mimic.
Dataset - 2026
UCSF RMaC
A multiphase renal mass dataset with pathology and up to three contrast-enhanced CT phases.
Best for
Subtype and enhancement-driven engineering questions where phase context matters more than a single static image.
Watch out
It is broader than pure RCC and still depends on careful downstream review of pathology, protocol variation, and label scope.
This map is meant to show the shape of the RCC literature rather than bury it inside a flat reading list. The top half is dominated by papers that explain how radiologists move from a generic enhancing renal mass to a more specific subtype or mimic interpretation. The bottom half shifts toward engineering resources, where the question is less "what is the diagnosis?" and more "what labels, phases, and tasks does this dataset actually give me?"
The biggest pattern is that the literature is much better at explaining enhancement behavior and subtype tendencies than at solving the benign-solid mimic problem cleanly. That is why the map includes both a subtype paper and a dedicated RCC-versus-oncocytoma validation study. It is also why the engineer path ends in KiTS23 and UCSF RMaC rather than a magical single RCC benchmark. That summary is an EnginRad inference from the cited research map, not a direct claim from one paper.4978
If you want the fastest reading path, start with the ACR white paper for the enhancement and incidental-mass frame, then the multimodality RCC review for subtype visuals, and then the small renal mass review for the actual mimic problem with AML and oncocytoma.123
Clinical and pathology background
RCC is a malignant epithelial renal tumor family, not a single look-alike mass. The major adult subtypes are clear cell, papillary, and chromophobe RCC, and radiologists care about subtype because the tumors do not all enhance or look the same.2 This is why an incidental "renal mass" discussion often becomes a phase-based conversation rather than a simple yes-or-no cancer label.
Clear cell RCC is the classic hypervascular subtype. It often enhances similarly to cortex in the corticomedullary phase, can look heterogeneous, and is more likely to show necrosis or venous invasion in aggressive cases.2 Papillary RCC usually sits at the opposite end of the spectrum: more hypovascular, often more homogeneous when small, frequently T2-dark, and sometimes hyperattenuating before contrast, which is why it can drift toward cyst or AML confusion.25 Chromophobe RCC often falls in between, with more intermediate enhancement and enough overlap with oncocytoma that imaging may stay suggestive rather than pathology-level definitive.24
Benign solid mimics overlap for different reasons. AML becomes easier when macroscopic fat is visible, but fat-poor AML can be T2-dark and enhancing without a clean fat clue. Oncocytoma is benign but may still be hypervascular, well-defined, and scar-forming, which is why it keeps showing up in the differential and in resected benign masses.53 That overlap is a major reason imaging sometimes stops at likely subtype or suspicious for RCC rather than pretending it can always replace pathology.
The management context matters too. For incidental renal masses, enhancement assessment, lesion size, and whether benign fat is present all shape the next step. MRI is often preferred for suspected small hypovascular masses, and a meaningful minority of the very smallest solid masses are still benign.1 So the deeper clinical background is not just "RCC versus not RCC." It is a workflow where radiologists balance subtype pattern recognition, benign mimic clues, and the limits of what imaging alone can prove.
Scope and caution
This page is educational and intentionally simplified for technical readers. It does not replace formal radiology interpretation, pathology review, urology evaluation, local policy, or patient-specific medical advice.
References
- Management of the Incidental Renal Mass on CT: A White Paper of the ACR Incidental Findings Committee: https://geiselmed.dartmouth.edu/radiology/wp-content/uploads/sites/47/2019/04/ACR_Renal2017.pdf
- A Concise Review of the Multimodality Imaging Features of Renal Cell Carcinoma: https://pmc.ncbi.nlm.nih.gov/articles/PMC7946646/
- The Role of CT Imaging in Characterization of Small Renal Masses: https://pmc.ncbi.nlm.nih.gov/articles/PMC9914376/
- Clear Cell Renal Cell Carcinoma: Discrimination from Other Renal Cell Carcinoma Subtypes and Oncocytoma at Multiphasic Multidetector CT: https://pubmed.ncbi.nlm.nih.gov/23382290/
- The different faces of renal angiomyolipomas on radiologic imaging: a pictorial review: https://pmc.ncbi.nlm.nih.gov/articles/PMC5965995/
- Artificial intelligence and radiomics in evaluation of kidney lesions: a comprehensive literature review: https://pmc.ncbi.nlm.nih.gov/articles/PMC10126666/
- KiTS23 challenge: https://kits-challenge.org/kits23/
- UCSF RMaC: University of California San Francisco 3D Multi-Phase Renal Mass CT Dataset with Tumor Segmentations: https://pubmed.ncbi.nlm.nih.gov/41728332/
- Usefulness of multidetector computed tomography to differentiate between renal cell carcinoma and oncocytoma. A model validation: https://pmc.ncbi.nlm.nih.gov/articles/PMC8765797/